Introduction
PSE for Stroke Recovery: Scientific Background
TNF
Etanercept
Stroke may result in widespread, sustained microglial activation in the brain
Etanercept reduces neuroinflammation by reducing microglial activation and neutralizing excess TNF
-
By attenuation of microglial activation through a direct effect on microglia and by interruption of the known microglia/TNF autocrine feed-forward loop[7,9,13,57,60,84-96]; and
-
By binding to soluble TNF and thereby reducing its biological activity [97,98].
Known physiological mechanisms are consistent with the rapid and sustained neurological improvement
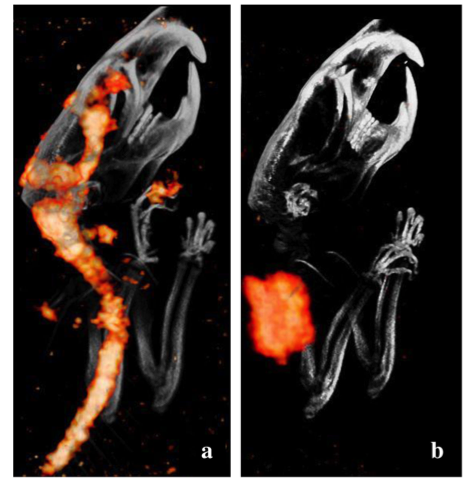
The Cerebrospinal Venous System
Perispinal injection enables rapid delivery of drugs to the brain via the CSVS